In August 2016, physicians at a hospital in Reno, Nevada, faced a scenario that infectious disease specialists had long warned about but hoped never to see. A woman in her seventies had been admitted with a severe bacterial infection after receiving medical treatment overseas. Doctors attempted antibiotic after antibiotic, moving through the entire arsenal available in the United States.
None worked.
Laboratory analysis revealed that the Klebsiella pneumoniae strain infecting her carried resistance mechanisms that rendered every available antimicrobial drug ineffective. The infection proved untreatable, and the patient ultimately died of septic shock.¹ Further testing confirmed that the organism was resistant to all 26 antibiotics examined, including drugs typically reserved as last-line treatments.²
Cases like this were once considered extreme anomalies. Now, antimicrobial resistance (AMR) is widely recognized as one of the most significant global public health challenges of the twenty-first century.³ A comprehensive global analysis estimated that drug-resistant bacterial infections were responsible for 1.27 million deaths worldwide in 2019, while 4.95 million deaths were associated with resistant infections.⁴ These numbers place AMR among the leading causes of death globally and underscore a rapidly growing threat to modern medicine.
Antibiotics transformed healthcare in the twentieth century, turning infections that once killed millions into treatable conditions. That foundation, however, is beginning to weaken.
The Biology of Resistance
Antibiotics work by targeting essential biological processes inside bacterial cells. Some disrupt the synthesis of bacterial cell walls, causing cells to rupture. Others interfere with protein production or DNA replication, preventing bacteria from multiplying.
But bacterial populations evolve quickly. When exposed to antibiotics, most bacteria die, yet some may possess genetic traits that allow them to survive. Those survivors reproduce, passing their resistance traits to future generations.
Over time, this evolutionary process allows resistant strains to dominate microbial populations exposed to antibiotics.
Bacteria have evolved numerous strategies to evade antimicrobial drugs. Some produce enzymes that break down antibiotics before they reach their targets. Others modify the molecular structures that antibiotics are designed to attack. Still others actively pump antimicrobial compounds out of the cell before they reach lethal concentrations.
And as if family-line mutations weren’t enough of a challenge, resistance traits can also spread rapidly within microbial communities. Bacteria can acquire resistance genes through mutation or by mechanisms that enable them to obtain DNA from neighboring microbes. This capacity rapidly accelerates the spread of resistance traits across bacterial populations and environments, especially in settings where multiple ill patients are in close proximity (e.g., hospitals, nursing homes).
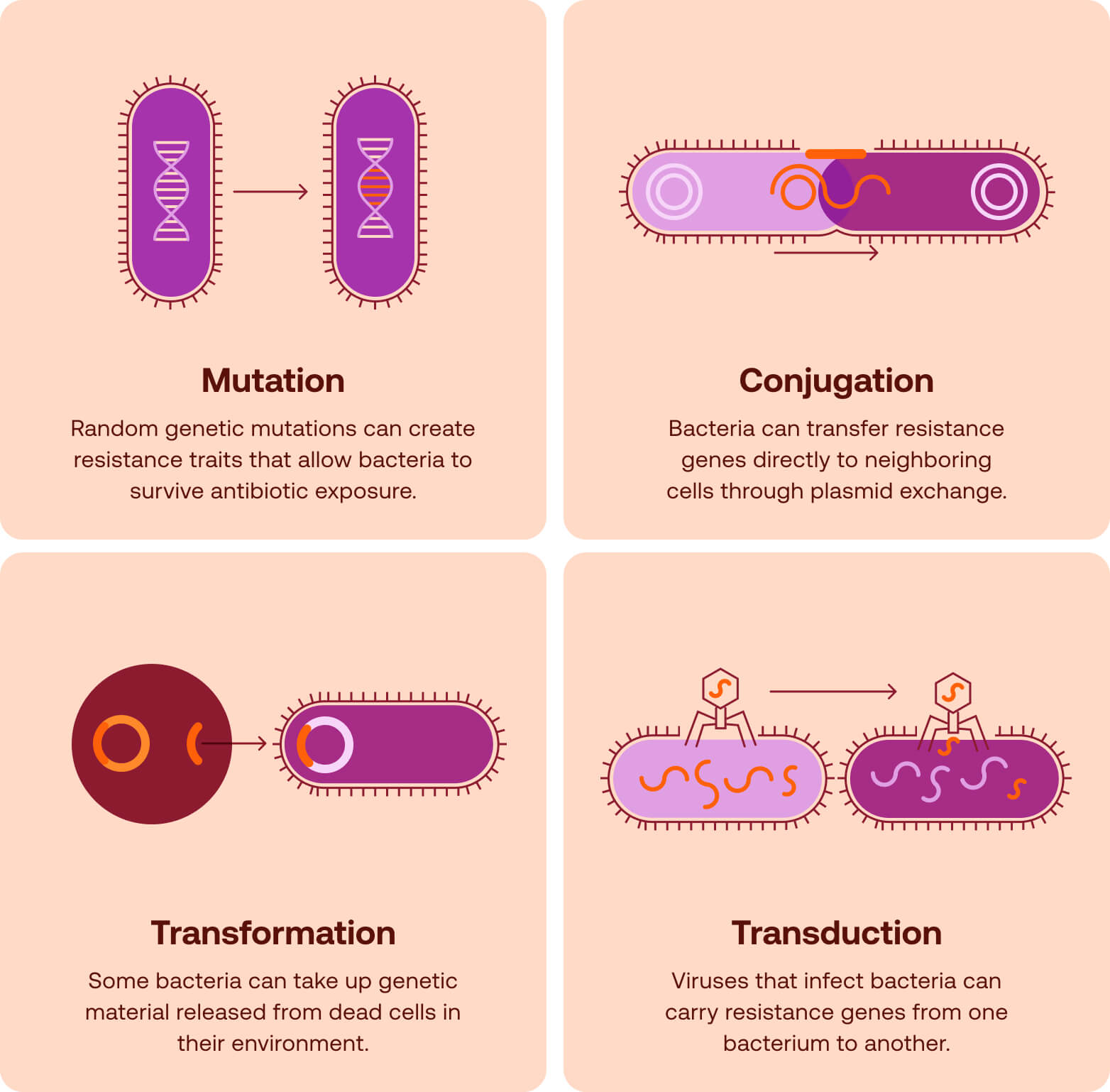
The result of a microbe’s proclivity for mutation is a continuous evolutionary arms race between microbes and medicine. Each new antibiotic exerts selective pressure that favors organisms capable of surviving it. As these organisms proliferate, treatments that once worked reliably become progressively less effective.
The Quiet Erosion of Modern Medicine
The consequences of antimicrobial resistance extend far beyond microbiology laboratories. Many of the medical procedures that define modern healthcare depend on antibiotics functioning reliably.
Routine surgeries rely on prophylactic antibiotics to prevent infection during or after procedures. Chemotherapy suppresses immune function, leaving patients dependent on antimicrobial drugs to prevent life-threatening infections. Organ transplantation requires long-term immunosuppression, which similarly increases vulnerability to bacterial disease.
When antibiotics fail, these procedures become significantly more dangerous.
Hospital-acquired infections caused by resistant organisms already pose serious challenges in healthcare systems worldwide. Pathogens such as Escherichia coli, Klebsiella pneumoniae, Staphylococcus aureus, and Acinetobacter baumannii account for a substantial share of global deaths linked to antimicrobial resistance.⁵
Infections caused by these organisms often require prolonged hospitalization and complex treatment regimens. When first-line antibiotics fail, clinicians may be forced to rely on older drugs that are less effective or more toxic.
This quiet erosion of antibiotic effectiveness rarely makes headlines, yet it is gradually reshaping everyday medical practice.
A Post-Antibiotic Era
Public health experts frequently warn of the possibility of a “post-antibiotic era,” a future in which common infections once again become difficult or impossible to treat.
The phrase may sound dramatic, but it reflects a trajectory already visible in isolated cases.
The Nevada infection illustrates the point starkly. The bacterium responsible for the patient’s illness carried resistance mechanisms that neutralized every antimicrobial drug available to physicians.¹
Although infections that resist all available antibiotics remain uncommon, they represent the logical endpoint of the evolutionary processes driving antimicrobial resistance.
If such pathogens become widespread, the implications for modern medicine could be profound.
Surgical procedures could become far more dangerous due to increased infection risk. Cancer treatments that suppress immune function could expose patients to infections that medicine can no longer control. Organ transplantation programs could face severe limitations because immunosuppressed patients would be highly vulnerable to untreatable infections.
Even infections that were once considered routine, including pneumonia or urinary tract infections, could again become major causes of death.
Modeling studies suggest that antimicrobial resistance could cause tens of millions of deaths globally in the coming decades if current trends continue without significant intervention.⁶
These projections underscore the urgency of addressing AMR before such scenarios become commonplace.
Why Resistance Is Accelerating
Several forces have contributed to the rapid emergence and spread of antimicrobial resistance over the past several decades.
One major driver is the widespread use of antibiotics in human medicine. While these drugs are essential tools for treating bacterial infections, they are sometimes prescribed unnecessarily, such as for viral illnesses where they offer no benefit.
Incomplete treatment courses can also contribute to resistance. When patients stop taking antibiotics before the full course is complete, partially resistant bacteria may survive and multiply.
Healthcare environments themselves can facilitate the spread of resistant organisms. Hospitals concentrate vulnerable patients, invasive procedures, and antimicrobial drugs in a single setting, creating conditions that allow resistant pathogens to emerge and spread.
Environmental factors may also play a role. Antibiotics released into wastewater from healthcare facilities, agriculture, or pharmaceutical manufacturing can expose environmental bacteria to low levels of antimicrobial compounds, encouraging the development of resistance.
Together, these pressures create a global microbial ecosystem in which resistant bacteria can thrive.
The Global Response
In recognition of the dire concern that scientists, public health officials, and other leaders have about the effects of antimicrobial resistance on our future, significant efforts are underway to combat AMR.
International health organizations and national public health agencies have established surveillance systems to monitor resistance patterns worldwide. These systems collect data on resistant infections, helping researchers identify emerging threats and guide policy responses.
Hospitals and healthcare systems are also increasingly implementing antimicrobial stewardship programs. These initiatives aim to ensure that antibiotics are used only when necessary and that the most appropriate drugs are selected for each infection.
Researchers and biotechnology companies are simultaneously working to develop new antimicrobial therapies. However, antibiotic development faces unique economic challenges. Unlike many other drugs, antibiotics are typically used for short treatment courses and are often reserved for severe infections, limiting their commercial revenue generation opportunities.
To address this issue, new funding models and public-private partnerships have emerged to support antimicrobial research and development.
Scientists are also exploring alternative approaches to combating bacterial infections, including bacteriophage therapies, antimicrobial peptides, and treatments that neutralize bacterial virulence factors rather than directly killing the organisms.
Advances in diagnostic technology are also helping clinicians identify infections more quickly and determine which antibiotics are most likely to be effective, reducing unnecessary antibiotic use and enabling more targeted treatments.
An Evolutionary Arms Race
Despite the role of modern medicine and other global industries in exacerbating the AMR crisis, antimicrobial resistance ultimately stems from evolutionary biology. Microorganisms evolve rapidly, and the selective pressures created by widespread antibiotic use ensure that resistance will continue to emerge.
The challenge facing global health systems is therefore not to eliminate resistance entirely, but to manage it. Doing so will require a combination of scientific innovation, responsible antibiotic use, improved infection prevention, and coordinated global action.
Vaccination programs, sanitation improvements, and infection control measures can all reduce the number of infections that require antibiotic treatment in the first place. Meanwhile, new antimicrobial therapies and diagnostics will be needed to keep pace with evolving pathogens.
The microbes humanity is fighting have had billions of years to refine their survival strategies. Antibiotics have been part of human medicine for barely a century.
Whether the next century belongs to resistant pathogens or to a new generation of antimicrobial innovation will depend on how effectively science, healthcare systems, and global policy adapt to this rapidly evolving challenge. disAMR is dedicated to fostering exposure, funding, collaboration, and persistence necessary to counter antimicrobial resistance. Will you join us? Add your name to the list to receive updates and information.